|
1/17/2024 0 Comments Moles to grams calculator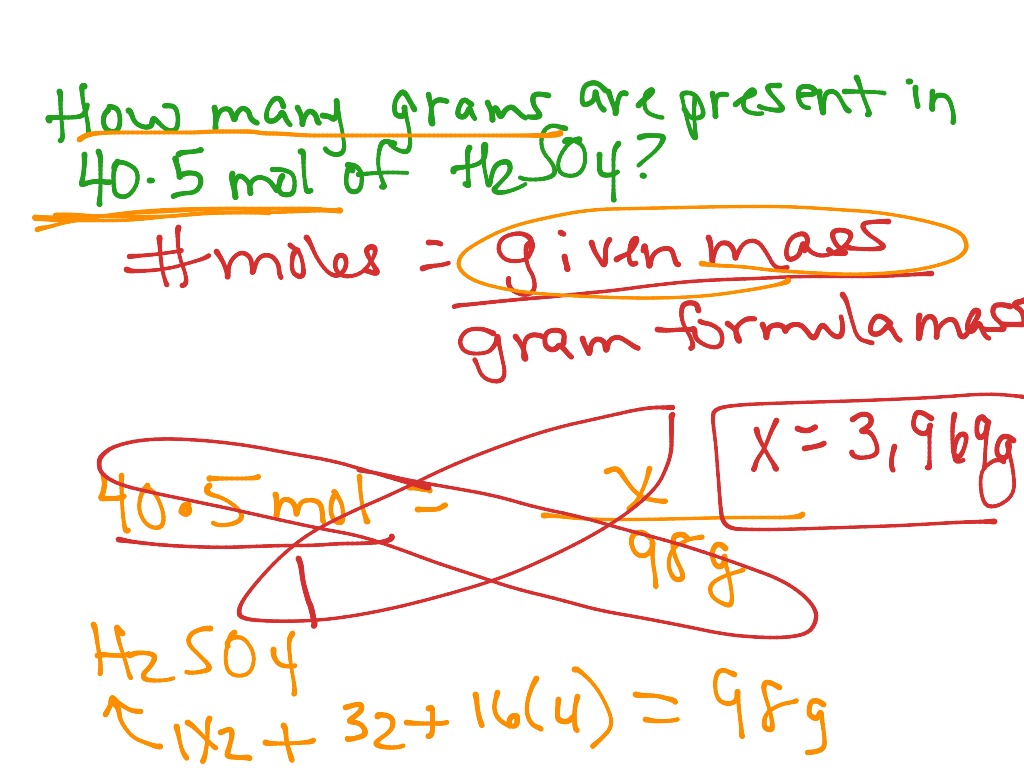
Question: How many moles are in 36 grams of water (H2O)? Calculate: Click the calculate button, and the calculator will provide you with the number of moles.Input values: Enter the mass of the substance (in grams) and its molar mass (in g/mol) into the Mol Calculator.This can usually be found in chemical reference materials or online resources. Find the molar mass: Look up the molar mass of the substance.You should know its chemical formula and the mass of the sample. Identify the substance: Determine the substance for which you want to calculate the moles.Here’s a step-by-step guide on how to use it: The Mol Calculator simplifies the process of calculating moles. It’s calculated by adding up the atomic masses of all the atoms in a molecule, taking into account the molecular formula. Molar Mass (g/mol) is the mass of one mole of that substance.Mass (g) is the mass of the substance in grams.Moles (mol) represent the quantity of a substance.Moles (mol) = Mass (g) / Molar Mass (g/mol) The formula to calculate moles is straightforward: This number is known as Avogadro’s number, which is approximately 6.022 x 10^23 entities/mol. A mole (abbreviated as ‘mol’) is defined as the amount of substance that contains the same number of entities (atoms, molecules, ions, or any other chemical entities) as there are in 12 grams of carbon-12. Formula:īefore delving into the calculator itself, it’s essential to understand the formula behind moles. In this article, we will explore what the Mol Calculator is, its formula, how to use it, provide an example, address common questions, and conclude with its significance in the world of chemistry. 
The Mol Calculator, also known as the moles calculator, is a valuable tool for students, researchers, and anyone interested in chemistry. One fundamental concept in chemistry is the mole, which is a unit of measurement used to count atoms and molecules. Our atoms to moles calculator easily converts atoms or molecules to moles.Chemistry is a subject that plays a crucial role in understanding the world around us, from the composition of substances to the reactions that shape our daily lives. The quantity of substance in moles is equal to the number of molecules divided by the Avogadro constant (6.02214076 × 10 23). The formula for converting molecules to moles is a little different than mass and volume because both molecules and moles are measures of quantity: You can also use our liters to moles calculator for this conversion. 
This convention is used for most calculations involving gasses and is intended to represent conditions at sea level. This relationship between liters and moles holds true at standard temperature and pressure (STP) since both temperature and pressure affect the volume of a gas. The quantity of a substance in moles is equal to the volume in liters of the ideal gas divided by 22.4 L/mol. You’ll still use a formula, but you do not need the molar mass of the gas for the conversion. The process for converting the volume of an ideal gas to moles is a little different than for mass. You can use the calculator above to calculate this more easily or even our grams to moles calculator to quickly convert grams to moles and moles back to mass. So, in this example, the quantity of 120 grams of aluminum oxide equals 1.18 moles. Note that the molar mass of aluminum oxide is 101.96 g/mol. Therefore, you need to be able to convert from weight and volume to moles and vice versa to conduct experiments in a laboratory or carry out reactions to manufacture products in industry.įor example, let’s calculate the quantity in moles equal to 120 grams of aluminum oxide, Al 2O 3. Using moles in chemical equations shows exactly how the reactants transform to make the products. However, most chemical equations are written in terms of moles so that the number of atoms of each type balances out on both sides of the equation. This conversion is useful because most chemicals are stored and measured either by weight (g) or volume (L). Below we’ll also cover how to convert volume and molecules to moles as well. Follow along, and we’ll show you how to convert mass to moles in just a few steps. You might be wondering how you can calculate the number of moles given a mass, volume, or amount of substance. Since the mole is a measure of quantity, all substances have the same number of atoms, molecules, and other particles in one mole of the substance. Because it’s such a large number, scientific notation is almost always used to represent it. This numerical value is known as the Avogadro constant or Avogadro’s number. One mole is equal to 6.02214076 × 10 23 elementary entities, such as atoms, molecules, electrons, or ions. It’s used as a standard measurement for a very large quantity of very small units of matter. 
The mole, abbreviated mol, is the SI base unit to measure the amount or quantity of a substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
